For example, we detected the IR light emitted when electrons in gaseous Ar-Xe mixtures are multiplicated near a thin wire (as in a wire chamber).
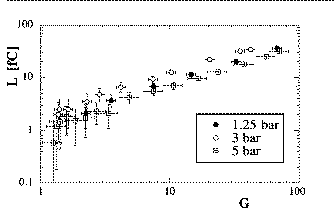 IR Light yield
L as
a function of the wire gain G in Ar-Xe gas at different gas
pressures.
IR Light yield
L as
a function of the wire gain G in Ar-Xe gas at different gas
pressures.
1. Introduction
The UV scintillation light from noble gases and liquids (Ar, Xe, Kr)
is well known for several decades. What is unknown is wheter those
substances emit also in the InfraRed (IR) region (let's say, at
wavelengths around or below 1 micron ).
We first discovered (G.Carugno,
NIMA 419 (1998), 617) that Argon gas scintillates in
the IR. A 5 MeV proton beam was sent in a cell containing Ar gas at 1 atm and room
temperature. The IR light was detected by a InGaAs diode cooled with
liquid nitrogen.
That result triggered a series of systematic studies to measure the
emission spectrum and the light yield and to verify if the IR emission
occurs in other gases and in the liquid state, too.
2. IR Emission under an Electric Field
We also discovered that the light emission intensity depends on the
electric field where the gas sits. We measured that the emission intensity
increases linearly with the electric field as soon as the field exceeds a
threshold value. We developed a naive model which explain the main
features of such emission (see
G. Bressi et al., Phys. Lett. A 280 (2001), 280)
Free electrons created by an ionizing particle are accelerated by the
electric field applied to the gas and acquire energy which can be
transferred to the gas atoms through inelastic collisions. Atoms are
therefore excited and emit IR light during their de-excitation.
We are now considering whether this "light amplification" can be
exploited for detection of particles.
For example, we detected the IR light emitted when electrons in
gaseous Ar-Xe mixtures are multiplicated near a thin wire (as in a wire
chamber).
 IR Light yield
L as
a function of the wire gain G in Ar-Xe gas at different gas
pressures.
IR Light yield
L as
a function of the wire gain G in Ar-Xe gas at different gas
pressures.
Below you find the list of our published papers related to these
researches.
The most direct way to understand IR scintillation is to measure the light
spectrum. IR photons in the wavelength range between 0.8 and 1.6 micron
are detected by means of solid-state detectors, namely, InGaAs.
The light is analyzed by a Michelson spectrometer. The medium under study
is excited by 100 keV electrons, accelerated by an electron gun.
Ar and Ne gas spectra are characterized by manu sharp lines whose
intensity is independent on the gas density. On the other hand, Xe
spectrum features a broad band that is red-shifted and broadened as gas
density is increased. The broad band is generated by transitions involving
the de-ectitation process of the Xe2 excimer. These transitions were
theoretically predicted but never observed so far.
The IR spectra of pure gas Xenon and gas Ar-Xe mixtures are measured and
explained in A.F.Borghesani et al., JCP
115(2001), 6042.
Liquid Argon and Xenon IR scintillate as well (G.Bressi et al., NIMA 440 (2000),
254).
4. IR light yield
We measured the light yield for a few gases using an alpha source. Among
investigated gases, Xe and Ar-Xe mixtures show the highest light yield,
about 2*10^4 photons/MeV. Note that this number is of the same order of
magnitude of NaI(Tl).
The measure is described in S.Belogurov et al., NIMA 452 (2000),
167.
On the other hand, the IR yield for liquid Xenon turned out to be much
smaller than for gas, at least in the region covered by the InGaAs
photodetector (G.Bressi et al., NIMA
461 (2001), 378). This is due to the pressure red-shift of the
emission line, as explained above.
5. Crystals
Many crystals scintillate in the IR, for example, the crystals used for lasers. Generally, however, the light output is poor and the scintillation time is long (microseconds to milliseconds) and therefore they hardly can be used for real detectors. We found CsI to be the best crystal among investigated ones. It scintillates at around 1.6 micron with a light yield of about 5*10^3 photons/MeV (S.Belogurov et al., NIMA 449 (2000), 254).
Here you find our papers related to this research.